
This is due to the fact that as you move down the periodic table, a new electron shell is added. The ionic radius of an element group (column) increases as you move from the top to the bottom of the column. The periodic table shows that the ionic radius and the atomic radius follow the same trends: The radius of an ion can be measured using x-ray crystallography or other techniques that are similar. A typical ionic radius ranges from 30 picometers (pm, which is equivalent to 0.3 Angstroms ) to 200 picometers (pm, which is equivalent to 2 Angstroms). Ionic radius values are difficult to obtain and tend to vary depending on the method used to determine the size of the ion being measured. An anion’s radius may be larger than the atomic radius. An anion has an extra electron, which increases the size of the electron cloud and may cause the ionic radius to be larger than the atomic radius. As a result of the removal of an electron, cations are typically smaller than neutral atoms because the remaining electrons are more tightly drawn in toward the nucleus of the atom. In accordance with the electric charge carried by the ion, the ionic radius can be either larger or smaller than the atomic radius (i.e., the radius of a neutral atom of an element). 
Because the boundary of an atom’s electron shell is somewhat ambiguous, the ions are frequently treated as if they were solid spheres fixed in a lattice. It is equal to half of the distance between two ions that are barely touching each other in space. 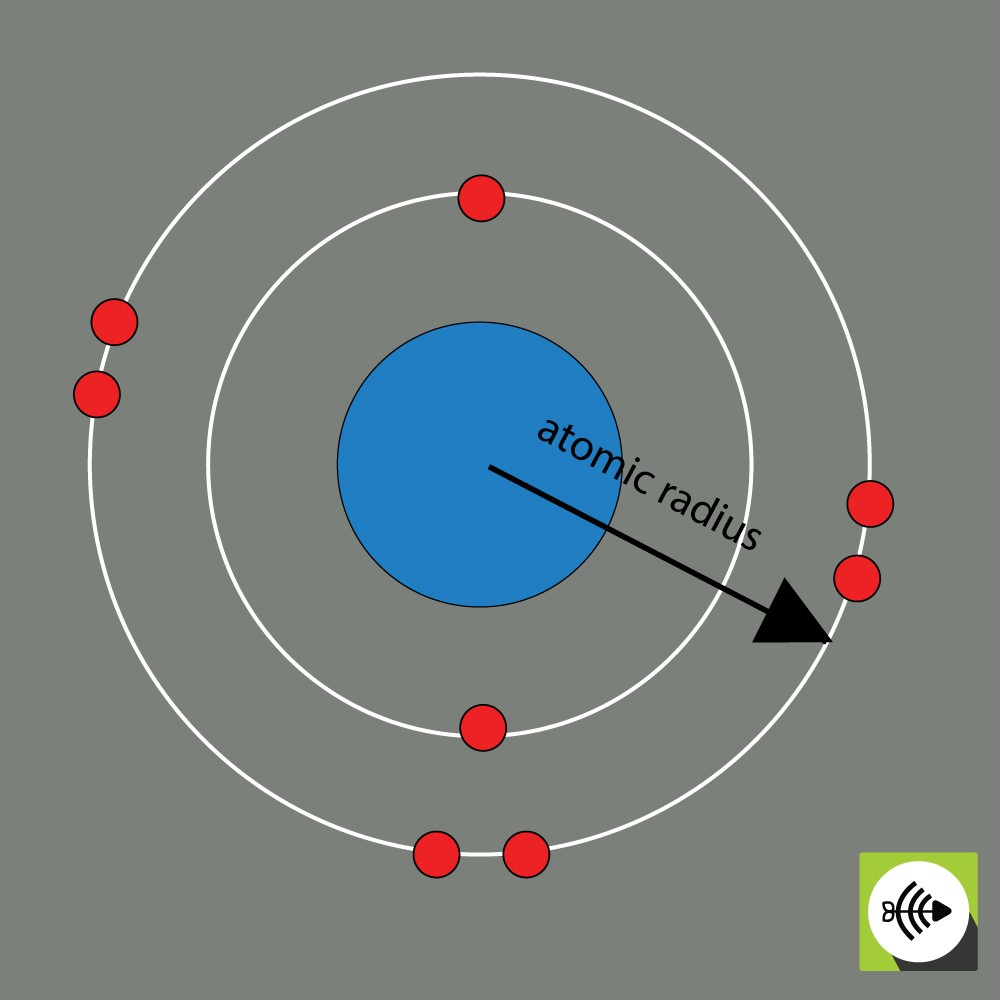
The ionic radius (plural: ionic radii) is a unit of measurement for the ionisation radius of an atom in a crystal lattice. This increase in radius can be explained by the presence of a greater number of subshells in the shell’s structure. In the course of progressing down the group, it is possible to observe an increase in the atomic and ionic radii of the elements. This is because the electron-electron repulsions cancel out the nuclear charge, resulting in a net neutral charge. Those placed between groups 7 and 10 have atomic radii that are roughly similar, whereas those placed between groups 11 and 12 have atomic radii that are bigger. 
My name is Walter Unglaub, and this what two factors determine the atomic radius of an element.In the transition elements from group 3 to group 6, the atomic and ionic radii of the transition elements drop due to the inadequate shielding provided by the small amount of d-electrons in the transition elements. So depending on the balance of the nuclear charge in the nucleus and the energy levels of the atom, this will determine the different sizes for different elements. However, if you have more electrons and more electron shells, then the size of the atom will tend to be larger compared to the effect of having a larger nuclear charge. So, the inner electrons will tend to be attracted more towards the nucleus. So, if we have a larger nuclear charge, meaning this nucleus is larger, there's going to be a stronger electromagnetic attraction between the positively charged protons and the negatively charged electrons. So, we might ask, how do these independently affect the size of an atom? Well, here we have an atom where we have a nucleus that is comprised of several neutrons and protons for example, and then if this is a neutral atom, we'll have as many electrons orbiting the nucleus as there are protons in the nucleus. Hello, my name is Walter Unglaub, and this is, "What two factors determine the atomic radius of an element?" Well there are several factors that determine the atomic size for any given element, but the two most important ones are the nuclear charge, which is given by the number of protons times the charge of a single electron, so this is a positive number, and the number of electron shells, or energy levels in the atom.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
